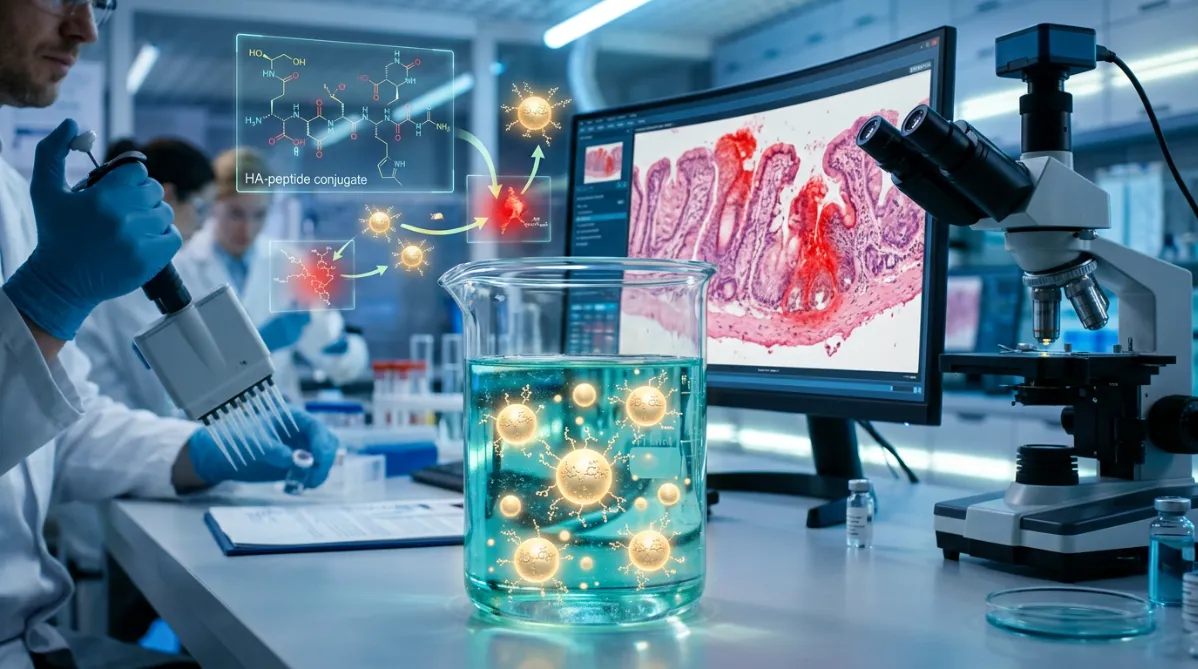
Breakthrough in Targeted Peptide Delivery
A groundbreaking new drug delivery system has emerged that could revolutionize the treatment of inflammatory bowel diseases like ulcerative colitis. Researchers at Georgia State University and Southwest University have successfully developed hyaluronic acid-functionalized nanoparticles that can precisely deliver therapeutic peptides to inflamed intestinal tissue, offering new hope for millions of patients worldwide.
The innovative system addresses one of the most persistent challenges in modern medicine: getting therapeutic compounds exactly where they need to go in the body while minimizing side effects. Traditional oral medications often suffer from poor absorption, rapid breakdown in the digestive system, and non-specific distribution throughout the body, leading to reduced efficacy and unwanted adverse effects.
The KPV Tripeptide: Nature's Anti-Inflammatory Agent
At the heart of this breakthrough is lysine-proline-valine (KPV), a naturally occurring tripeptide derived from the hormone α-melanocyte-stimulating hormone (α-MSH). This small but powerful molecule has demonstrated remarkable anti-inflammatory properties, making it an ideal candidate for treating inflammatory bowel diseases.
"KPV represents a significant advancement in peptide therapeutics," explains Dr. Sarah Mitchell, a gastroenterology specialist not involved in the study. "Unlike synthetic drugs that often come with significant side effects, this naturally derived compound works with the body's own anti-inflammatory pathways."
The tripeptide functions by modulating key inflammatory signaling pathways, including the nuclear factor-kappa B (NF-κB) pathway, which plays a central role in inflammatory responses. When inflammation occurs in the intestinal tract, as seen in ulcerative colitis, KPV can help restore the delicate balance between pro-inflammatory and anti-inflammatory signals.
However, the therapeutic potential of KPV has been limited by its poor stability and rapid degradation when administered orally. Peptides are notoriously difficult to deliver effectively through the gastrointestinal tract due to the harsh acidic environment of the stomach and the presence of various digestive enzymes that break down proteins and peptides.
Hyaluronic Acid: The Perfect Carrier
The research team's solution involves encapsulating KPV within nanoparticles functionalized with hyaluronic acid (HA), a naturally occurring polymer found throughout the human body. Hyaluronic acid serves multiple crucial functions in this delivery system:
Targeted Recognition
Hyaluronic acid binds specifically to CD44 receptors, which are overexpressed on the surface of inflamed intestinal epithelial cells and immune cells involved in inflammatory bowel disease. This receptor-mediated targeting ensures that the therapeutic payload is delivered precisely where it's needed most.
Enhanced Stability
The HA coating protects the encapsulated KPV from degradation by digestive enzymes and the acidic stomach environment, allowing more of the active compound to reach its target destination intact.
Improved Bioavailability
The nanoparticle formulation enhances the absorption and bioavailability of KPV compared to free peptide administration, resulting in more effective therapeutic concentrations at the site of inflammation.
Mucoadhesive Properties
Hyaluronic acid exhibits mucoadhesive properties, meaning it can bind to the mucus layer lining the intestinal tract. This adhesion prolongs the residence time of the nanoparticles in the gut, allowing for sustained release of the therapeutic peptide.
Nanoparticle Engineering: Precision at the Molecular Level
The development of effective nanoparticle delivery systems requires precise engineering at the molecular level. The research team carefully optimized several critical parameters:
Particle Size and Distribution
The nanoparticles are designed to be approximately 100-200 nanometers in diameter, a size range that optimizes several factors:
- Small enough to penetrate the mucus barrier
- Large enough to avoid rapid clearance by the kidneys
- Optimal for cellular uptake through endocytosis
Surface Charge
The surface charge of the nanoparticles affects their interaction with biological membranes and their stability in biological fluids. The HA functionalization provides an optimal surface charge that promotes cellular uptake while maintaining stability.
Drug Loading Efficiency
The researchers achieved high drug loading efficiency, ensuring that each nanoparticle carries a therapeutically relevant amount of KPV. This efficiency is crucial for minimizing the total dose required and reducing potential side effects.
Release Kinetics
The nanoparticles are designed to provide controlled release of KPV over an extended period, maintaining therapeutic concentrations at the target site while minimizing systemic exposure.
Clinical Implications for Ulcerative Colitis Treatment
Ulcerative colitis affects approximately 1 million people in the United States alone, causing chronic inflammation of the large intestine that leads to symptoms including bloody diarrhea, abdominal pain, and urgent bowel movements. Current treatments include:
Conventional Therapies
- 5-Aminosalicylates (5-ASAs): Anti-inflammatory drugs that are often first-line treatments but may not be effective for all patients
- Corticosteroids: Powerful anti-inflammatory medications that are effective but carry significant long-term side effects
- Immunosuppressants: Drugs that suppress the immune system but increase infection risk
- Biologics: Targeted therapies that are expensive and may lose effectiveness over time
Limitations of Current Approaches
Existing treatments often suffer from:
- Systemic side effects due to non-specific distribution
- Variable efficacy among patients
- Development of resistance or loss of response over time
- High cost, particularly for biologic therapies
- Need for frequent dosing or invasive administration routes
Advantages of the New Delivery System
The HA-functionalized nanoparticle system addresses many of these limitations:
Targeted Delivery: By specifically targeting inflamed tissue through CD44 receptors, the system minimizes exposure to healthy tissue and reduces side effects.
Enhanced Efficacy: Improved bioavailability and targeted delivery result in higher therapeutic concentrations at the site of inflammation.
Reduced Dosing Frequency: Sustained release properties may allow for less frequent dosing, improving patient compliance.
Cost-Effectiveness: Using naturally occurring compounds like KPV and HA could potentially reduce treatment costs compared to expensive biologic therapies.
Improved Safety Profile: The natural origin of both the therapeutic agent and the delivery system components suggests a favorable safety profile.
Experimental Validation and Results
The research team conducted comprehensive preclinical studies to evaluate the effectiveness of their delivery system. Key findings include:
In Vitro Studies
- Demonstrated specific binding to CD44 receptors on inflammatory cells
- Showed sustained release of KPV over 24-48 hours
- Confirmed anti-inflammatory activity in cell culture models
Animal Studies
- Significant reduction in colonic inflammation in mouse models of ulcerative colitis
- Improved histological scores compared to control treatments
- Reduced inflammatory markers including cytokines and chemokines
- Enhanced mucosal healing and restoration of epithelial barrier function
Safety Evaluation
- No significant adverse effects observed in treated animals
- Good tolerance of the nanoparticle formulation
- Minimal systemic exposure, confirming targeted delivery
Future Directions and Clinical Translation
The success of this nanoparticle delivery system opens several exciting avenues for future research and clinical development:
Expanded Peptide Library
The platform technology could be adapted to deliver other therapeutic peptides and proteins, potentially expanding treatment options for various inflammatory conditions.
Combination Therapies
The nanoparticles could be loaded with multiple therapeutic agents, allowing for combination therapy approaches that target different aspects of the inflammatory cascade.
Personalized Medicine
By modifying the surface functionalization or loading different therapeutic agents, the system could be customized for individual patients based on their specific disease characteristics.
Clinical Trial Design
The next crucial step involves designing and conducting clinical trials to evaluate the safety and efficacy of the system in human patients. This will require:
- Phase I safety studies in healthy volunteers
- Phase II proof-of-concept studies in ulcerative colitis patients
- Large-scale Phase III efficacy trials
Regulatory Considerations
The path to regulatory approval will need to address:
- Characterization of the nanoparticle formulation
- Manufacturing consistency and quality control
- Long-term safety evaluation
- Comparison with existing standard-of-care treatments
Broader Impact on Drug Delivery Science
This breakthrough represents more than just a new treatment for ulcerative colitis; it demonstrates the potential of sophisticated nanoparticle delivery systems to transform how we approach therapeutic intervention.
Paradigm Shift
The success of this approach signals a shift from systemic drug administration to precision-targeted therapy, potentially reducing side effects while improving therapeutic outcomes.
Technology Platform
The HA-functionalized nanoparticle platform could serve as a template for developing treatments for other inflammatory conditions, including:
- Crohn's disease
- Rheumatoid arthritis
- Inflammatory skin conditions
- Other autoimmune disorders
Economic Implications
Successful development of this technology could:
- Reduce healthcare costs by improving treatment efficacy
- Decrease hospitalizations and complications
- Provide more affordable alternatives to expensive biologic therapies
Challenges and Considerations
Despite the promising results, several challenges remain in translating this technology to clinical practice:
Manufacturing Scalability
Producing nanoparticles with consistent quality at commercial scale requires sophisticated manufacturing processes and quality control measures.
Long-term Safety
While initial safety data are encouraging, long-term effects of repeated nanoparticle administration need thorough evaluation.
Patient Variability
Differences in individual patient physiology, disease severity, and genetic factors may affect treatment response and require personalized dosing strategies.
Regulatory Approval
The complex nature of nanoparticle formulations presents unique regulatory challenges that may require new evaluation frameworks.
Conclusion
The development of hyaluronic acid-functionalized nanoparticles for targeted delivery of the KPV tripeptide represents a significant milestone in both peptide therapeutics and drug delivery science. By addressing the fundamental challenges of peptide stability, bioavailability, and targeted delivery, this innovation opens new possibilities for treating inflammatory bowel diseases and other inflammatory conditions.
The success of this approach demonstrates the power of combining natural therapeutic compounds with sophisticated delivery technologies to create more effective and safer treatments. As this technology moves toward clinical trials, it holds the promise of providing new hope for patients suffering from ulcerative colitis and potentially other inflammatory conditions.
The broader implications of this breakthrough extend beyond a single disease, offering a platform technology that could revolutionize how we deliver therapeutic peptides and proteins throughout the body. As personalized medicine continues to evolve, such targeted delivery systems will likely play an increasingly important role in optimizing therapeutic outcomes while minimizing adverse effects.
For the millions of patients worldwide struggling with inflammatory bowel diseases, this breakthrough represents not just scientific advancement, but the potential for significantly improved quality of life through more effective, targeted treatments. The journey from laboratory bench to patient bedside continues, but the foundation laid by this research provides a strong platform for future therapeutic innovations.